Avoid FOMO by catching #DiscoverBMB 2023’s greatest hits
Did you attend the Discover BMB 2023 spotlight session on drug screening and development, catch both of Squire Booker’s award lectures, or boost your education and professional development skills at the pedagogy-focused talks?
If you missed out on any of these über-popular experiences, you can still hone your knowledge by getting access to the 2023 talks on demand!
At a meeting like #DiscoverBMB, it’s impossible to see everything because of the wealth of information shared and exchanged by so many top scientists and educators.
Here are the top five most-viewed abstracts for talks you can watch today:
1. Discovering new molecular mechanisms of cell death
Abstract: Non-apoptotic cell death is important for human health and disease. The full complement of non-apoptotic processes that can be activated in mammalian cells is unclear. However, over the last decade we have uncovered several novel mechanisms of non-apoptotic cell death, including ferroptosis and an unusual form of palmitoylation-dependent cell death. I will describe our discovery of these cell death mechanisms and discuss how the application of small molecule probes, genetic screening, lipidomic analysis, and biochemical approaches is leading to new insights into the regulation of both processes. Disrupted lipid metabolism is emerging as one important mechanism governing non-apoptotic cell death and I will describe our efforts to isolate specific lipid metabolic enzymes important for the regulation of non-apoptotic cell death. I will also discuss recent progress in understanding how these non-apoptotic cell death mechanisms can be exploited to treat diseases such as cancer.
2. Revealing the secrets of metalloenzymes one snapshot at a time
Catherine Drennan, Massachusetts Institute of Technology and Howard Hughes Medical Institute, winner of the William C. Rose Award
Abstract: How do microbes live on the pollutant carbon monoxide? How do microbes split the triple bond of nitrogen gas? When it comes to performing difficult chemistry, microbes often combine a protein scaffold with a highly reactive metallocofactor, employing a hired gun, if you will. The Drennan lab combines X-ray crystallography and cryo-electron microscopy with other biophysical methods with the goal of revealing the secrets behind metalloenzyme chemistry. In her Rose Award presentation, Drennan will present her lab’s structural studies of ribonucleotide reductases (RNRs). RNRs employ metallocofactors to generate radical species to afford the conversion of ribonucleotides (the building blocks of RNA) to deoxyribonucleotides (the building blocks of DNA). These enzymes are chemotherapeutic targets and proposed antibacterial targets. Here, she will describe how the “resolution revolution” of cryo-electron microscopy allowed for the capture of an active state structure of ribonucleotide reductase for the first time.
3. How to build, manipulate and destroy functional nanodomains in cell membranes
Anne Kenworthy, University of Virginia School of Medicine, winner of the Mildred Cohn Award in Biological Chemistry
Abstract: While all membranes share a characteristic bilayer morphology, their lateral organization can be remarkably complex. In biological membranes, lipids and proteins self-assemble to generate a variety of compositionally distinct domains that range in size from nanometers to microns, exist over a wide range of time scales, and assume varying curvatures and morphologies. Our group seeks to understand the physicochemical principles that govern the assembly and function of two related yet distinct classes of membrane nanodomains: membrane rafts and caveolae. Both reside within the plasma membrane of cells, form in a cholesterol-dependent manner, regulate multiple cellular processes, and, when defective, contribute to human disease. Yet, they differ substantially in morphology, lifetime, and function. How these two discrete classes of domains accomplish the many tasks ascribed to them remains a fundamental unanswered question in biology. Here, I summarize lessons we have learned using biophysical approaches about the connections between the structure, dynamics and function of these enigmatic nanodomains. I also present recent insights emerging from our work on two major questions in the field: 1) What is the structural basis for caveolae formation? and 2) Are rafts druggable targets?
4. Adventures in the biological chemistry, molecular biology and evolution of sialic acids
Ajit Varki of the University of California San Diego, winner of the Herbert Tabor Research Award
Abstract: Apparently immutable laws of mathematics, physics and chemistry were co-opted by biological evolution via natural selection, generating all known life forms from a last universal common ancestor. Some of the evolutionarily more ancient aspects of biological chemistry (e.g., glycolysis) and molecular biology (e.g., the genetic code and the central dogma enunciated by Francis Crick that genetic information flows from DNA, to RNA, to protein, or RNA to protein) are highly conserved due to long-term purifying selection that (along with fractal patterns) gives the illusion of “design”. But, as Crick himself later opined, “there are no laws in biology, only gadgets”. Given that the genetic code represents generation of order out of chaos, it is not surprising that the ongoing inter-species interactions of rapidly evolving pathogens and symbionts with hosts has resulted in the emergence of less conserved structures and functional phenomena. Of all the macromolecules in nature, the most diverse and rapidly evolving are glycans, which are also the least studied and understood––effectively “the dark matter of the biological universe.” Among vertebrate glycans, some of the most diverse are sialic acids, which are prominently displayed at the outermost end of glycan chains in the deuterostome lineage of animals, and on pathogens and symbionts that interact with them. My group has spent many years studying the biological chemistry and molecular biology of sialic acids and sialoglycan-recognizing proteins in evolution, with a particular emphasis on human origins and disease. From the earliest days of my education in biology and medicine, the Journal of Biological Chemistry had a major impact on my intellectual development as a physician-scientist. In this lecture I will cite a few such examples, with some focus on instances where multiple (often back-to-back) papers in the JBC helped focus attention on functionally significant variations of sialic acids, their glycosidic linkages, modifications such as O-acetyl and N-glycolyl groups, free amines and sulfate esters, and various combinations thereof. I will also briefly mention my limited but truly memorable interactions with Herb Tabor in the early days of online and open-access publishing, and a failed attempt to establish a uniform code of ethics for editors of biomedical journals.
5. Antibodies against emerging infectious diseases
Erica Ollmann Saphire of the La Jolla Institute for Immunology, winner of the Bert and Natalie Vallee Award in Biomedical Science
Abstract: Antibodies provide a critical line of defense against infectious diseases and are a primary goal of protective vaccines. Antibodies themselves can also serve as therapeutics or as design templates to develop or improve needed vaccines. Understanding the structure, reactivity, breadth, and protective activities of antibodies is key to both therapeutic and vaccine design. We have galvanized global consortia to understand which antibodies are most effective against viruses like Ebola, Lassa, and SARS-CoV-2. These consortia analyzed which antibodies demonstrate potent neutralization, which inspire Fc-mediated effector functions, which exhibit breadth across strains, and which yield durable neutralization despite emergence of highly mutated variants, and why. Results from the Bill and Melinda Gates Foundation-, GHR- and NIAID-funded Coronavirus Immunotherapeutics Consortium (CoVIC) that studied 400 candidate therapeutic antibodies against SARS-CoV-2 afforded fine epitope mapping on the spike protein; these distinctions forecast antibody activities. Structures of CoVIC IgGs in complex with the spike explain why some antibodies retain potency even though the footprint they target is highly mutated, like in the Omicron Variants. The geometry of their IgG recognition allows bivalent binding that confers avidity to preserve neutralization activity. Results of another project, the Viral Immunotherapeutic Consortium (VIC), mapped first-in-class antibody therapies for Ebola Virus Disease (Inmazeb) and Lassa Fever (Arevirumab), explained their mechanism of action and why a cocktail approach that combines complementary functions endows resistance to escape and opens multiple avenues of in vivo protection. For Lassa virus in particular, these studies help explain why neutralizing antibodies against this virus are so rare and provide specific design strategies to develop antigens to find and elicit more potently neutralizing antibodies.
Popular speakers
Below are the top five most-searched-for speakers in the #DiscoverBMB app. Watch their talks.
1. Squire J. Booker of Pennsylvania State University, winner of the ASBMB Merck Award and the Ruth Kirschstein Diversity in Science Award
2. David Baker of the University of Washington, invited speaker for Artificial Intelligence and Machine Learning in Structural Biology, Drug Design and Systems Biology symposium
3. E. Dale Abel of the University of California, Los Angeles, invited speaker for Advances in Organismal and Cellular Metabolism symposium
4. Fangliang Zhang of the University of Miami Miller School of Medicine, co-organizers of the posttranslational modifications interest group
5. David M. Shechner of the University of Washington, invited speaker for the Regulation of RNA symposium

How to watch
You can purchase unlimited access to the recordings of all #DiscoverBMB 2023 talks here. Your purchase will allow you to access 11 award lectures, 35 symposia and 54 spotlight sessions! The recordings will feature speaker audio and slideshow presentation. You can purchase access for $299, and you’ll receive a special discount of $100 off if you are a member of the American Society of Biochemistry and Molecular Biology!
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Targeting Toxoplasma parasites and their protein accomplices
Researchers identify that a Toxoplasma gondii enzyme drives parasite's survival. Read more about this recent study from the Journal of Lipid Research.
Scavenger protein receptor aids the transport of lipoproteins
Scientists elucidated how two major splice variants of scavenger receptors affect cellular localization in endothelial cells. Read more about this recent study from the Journal of Lipid Research.
Fat cells are a culprit in osteoporosis
Scientists reveal that lipid transfer from bone marrow adipocytes to osteoblasts impairs bone formation by downregulating osteogenic proteins and inducing ferroptosis. Read more about this recent study from the Journal of Lipid Research.

Unraveling oncogenesis: What makes cancer tick?
Learn about the ASBMB 2025 symposium on oncogenic hubs: chromatin regulatory and transcriptional complexes in cancer.

Exploring lipid metabolism: A journey through time and innovation
Recent lipid metabolism research has unveiled critical insights into lipid–protein interactions, offering potential therapeutic targets for metabolic and neurodegenerative diseases. Check out the latest in lipid science at the ASBMB annual meeting.
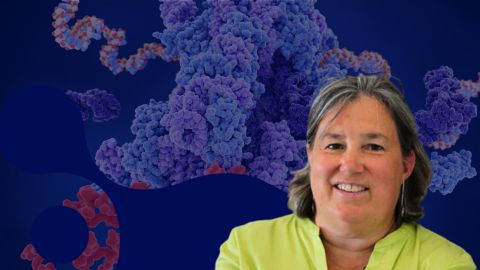
Melissa Moore to speak at ASBMB 2025
Richard Silverman and Melissa Moore are the featured speakers at the ASBMB annual meeting to be held April 12-15 in Chicago.






