From the journals: MCP
We offer a selection of papers on a variety of topics recently published in the journal Molecular & Cellular Proteomics.
Using co-elution to study protein interactions
Studying how the vast network of proteins and molecules in a cell interact, a discipline known as interactomics, is crucial to researchers’ fundamental understanding of biological processes and for the development of new medicines and biotechnology. Among the technical strategies for mapping the interactome, co-elution is a global protein interaction mapping method. However, strategies vary across studies that utilize co-elution, depending on experimental considerations.
In a review in the journal Molecular & Cellular Proteomics, Daniela Salas and colleagues at the University of British Columbia delineate co-elution methods used to map protein–protein interaction networks and discuss important considerations in designing co-elution studies, such as the choice of separation method and how to analyze co-elution profiling studies. The researchers also discuss the benefits of co-elution versus other mapping methods, including the time and resources required to perform the protein mapping and the number of protein interactions that can be explored.
Confident identification of citrullinated peptides
Citrulline is an amino acid not encoded in the genome. It is generated by a post-translational modification to the amino acid arginine, a process known as citrullination. In recent years, scientists concerned with the immune system have been paying attention to citrullination because of its role in inducing anti-citrullinated proteins/peptide antibodies, which results in an autoimmune reaction where the host’s immune system attacks its healthy tissue. The bacteria Porphyromonas gingivalis generates citrullinated epitopes in the periodontium, which contributes to chronic periodontitis and recently has been linked to rheumatoid arthritis.
Using a new two-dimensional heptafluorobutyric acid–based separation system combined with liquid chromatography–mass spectrometry, Daniel Larsen and colleagues at the University of Southern Denmark analyzed the outer membrane vesicles and other related elements of P. gingivalis to identify 79 citrullinated proteins with 161 citrullination sites. These results were reported in a paper published in the journal Molecular & Cellular Proteomics. This work establishes a method for identifying citrullinated proteins that will advance development of treatments for human autoimmune and inflammatory diseases.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
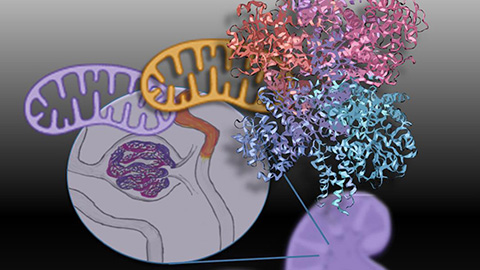
Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.
Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.
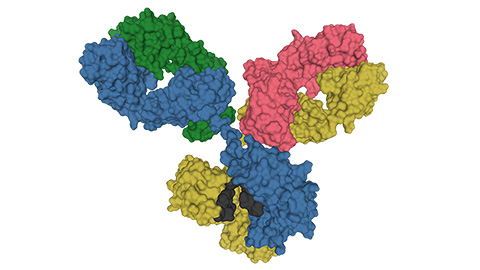
Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.
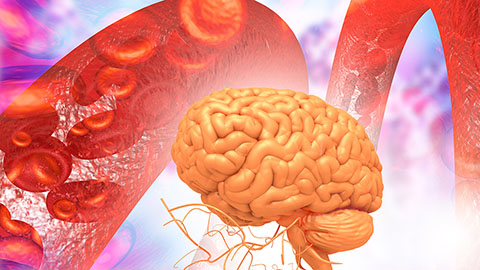
Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.
Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

